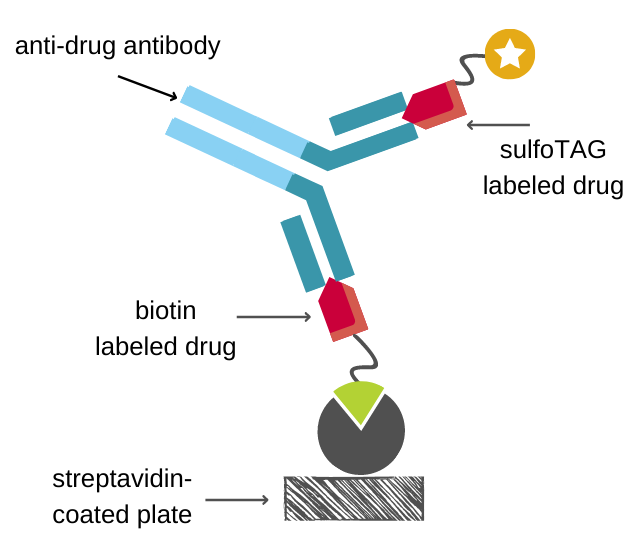
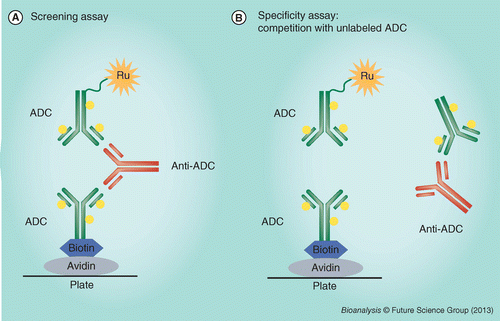
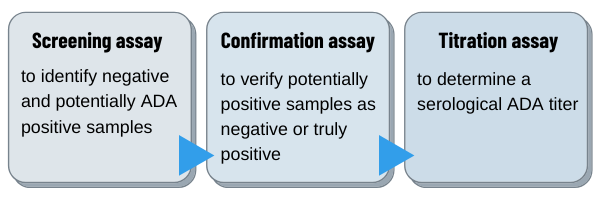
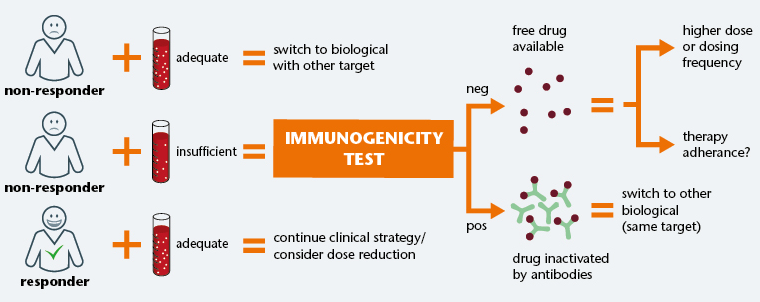
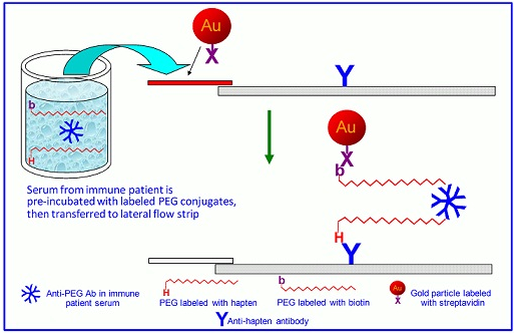
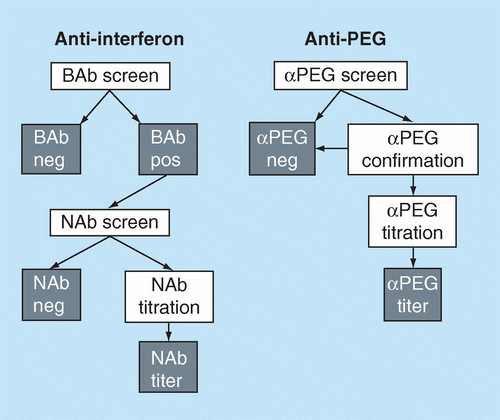
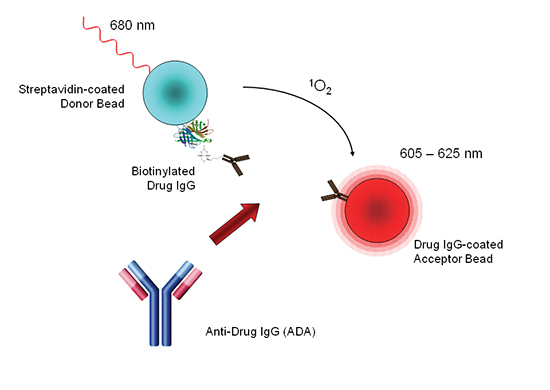
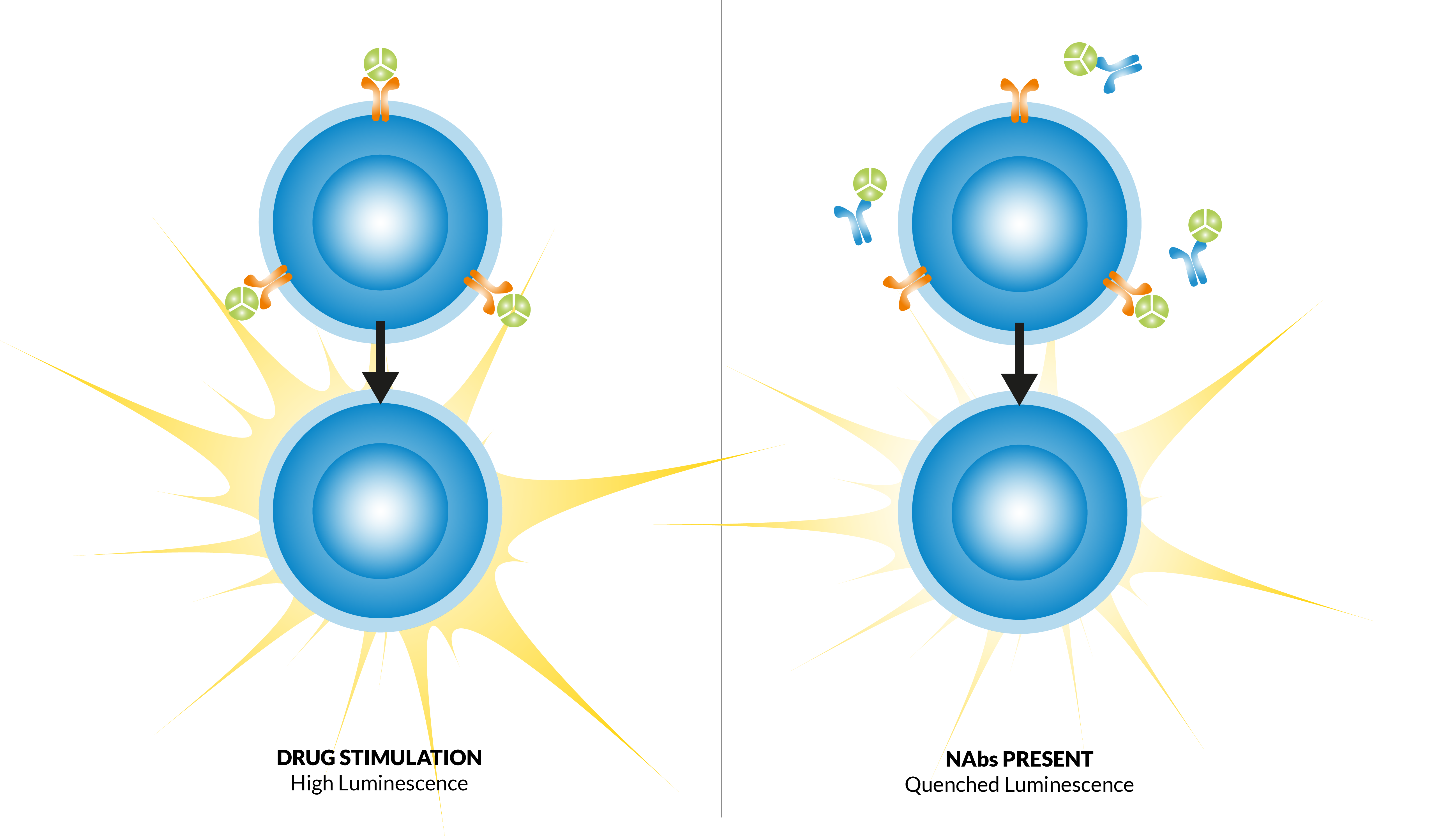
Veeda Clinical Research - "Immunogenicity Testing of Therapeutic Protein Products—Developing and Validating Assays for Anti-Drug Antibody Detection" represents current FDA thinking about developing and validating assays for anti-drug antibody (ADA ...
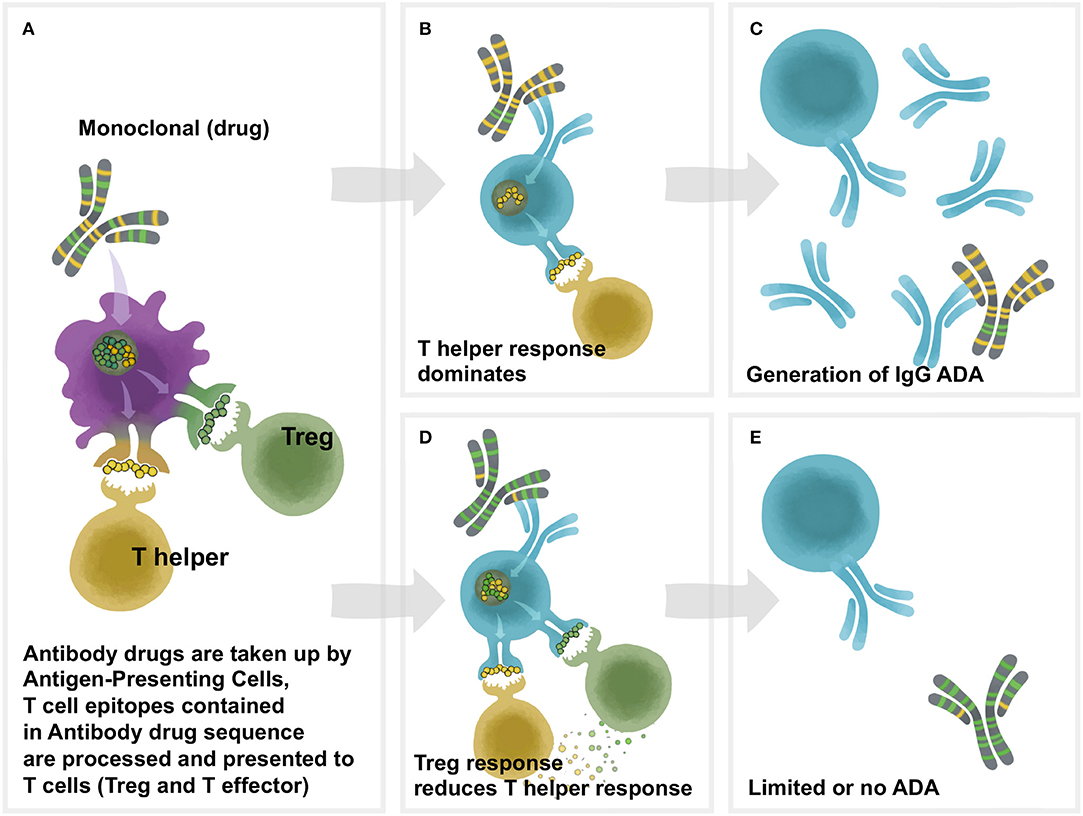
Frontiers | T-Cell Dependent Immunogenicity of Protein Therapeutics Pre-clinical Assessment and Mitigation–Updated Consensus and Review 2020 | Immunology

Immunogenicity assessment of biotherapeutic products: An overview of assays and their utility - ScienceDirect
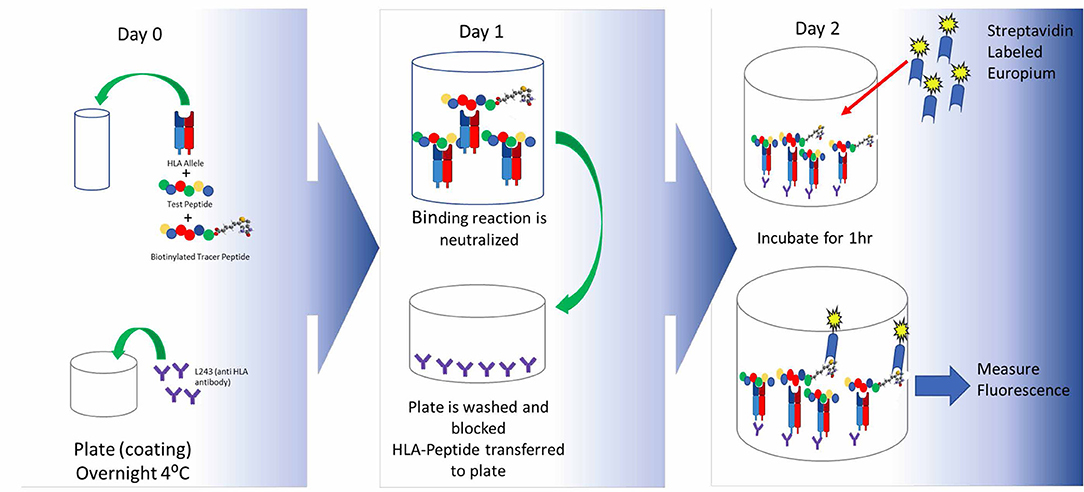
Frontiers | T-Cell Dependent Immunogenicity of Protein Therapeutics Pre-clinical Assessment and Mitigation–Updated Consensus and Review 2020 | Immunology