The Language of Clinical Trials. Objectives Objectives: At the conclusion of this discussion, participants will be able to: –Define clinical research. - ppt download

Principal Investigator Training - Role of Principal Investigator in Clinical Research — Clinical Research Certification
PLOS ONE: Workflow in Clinical Trial Sites & Its Association with Near Miss Events for Data Quality: Ethnographic, Workflow & Systems Simulation
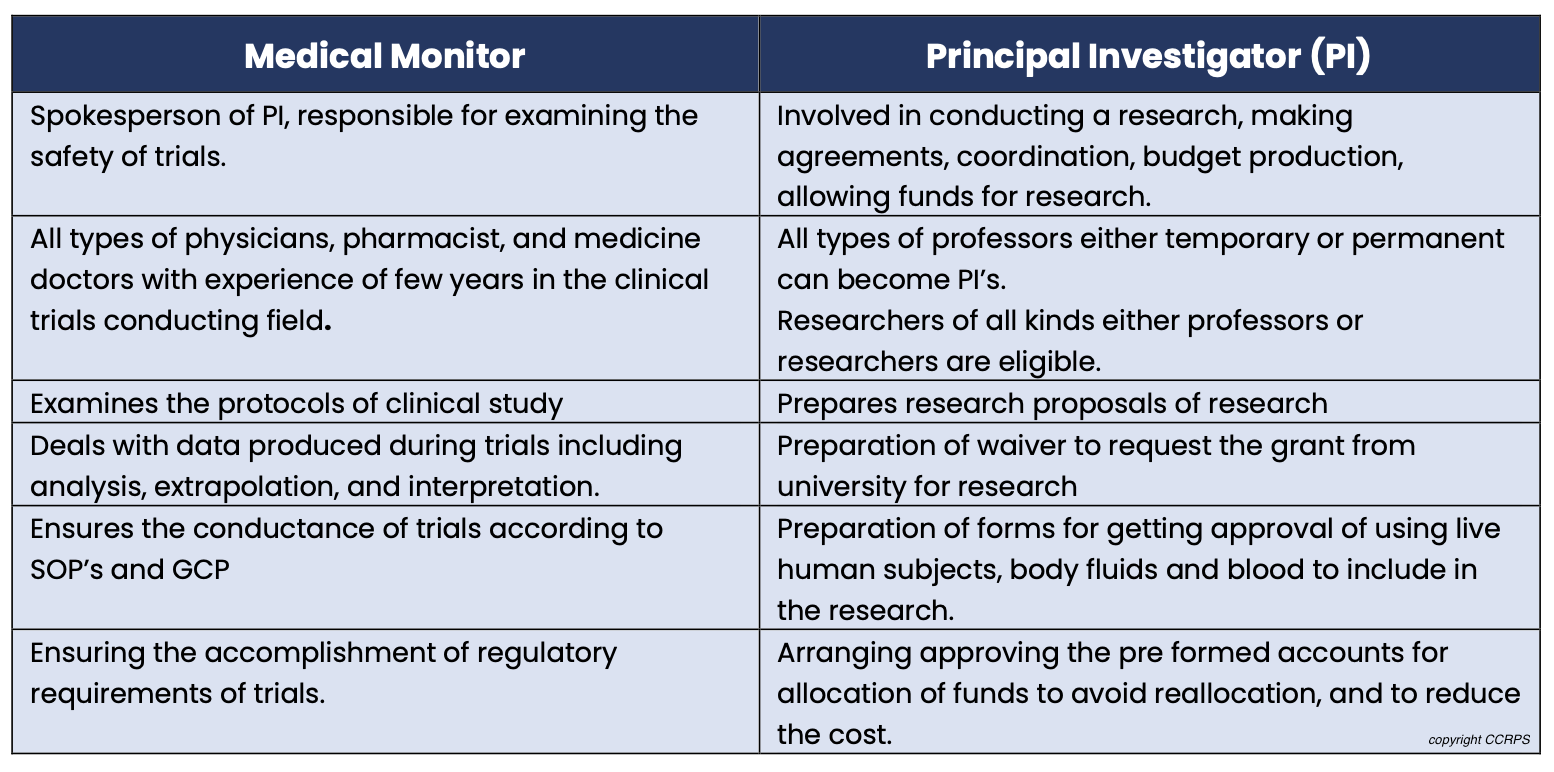
Medical Monitoring in Clinical Research - Non Clinical Physician Jobs — Clinical Research Certification

Principal Investigator Training - Role of Principal Investigator in Clinical Research — Clinical Research Certification